Mars, Super-duper Fast Charging, Salty & Liquid Metal Batteries: New Tech to Charge the World
Beyond Lithium-Ion Batteries - Part 2
Another month goes by and here we are with another edition of the Green Innovations Newsletter. Welcome back. We are diving back in to the world of battery technology to see what lies beyond Lithium-Ion horizon.
We’ll explore batteries that can be recharged faster than filling petrol at the pump, look at how technology built for Mars is driving battery tech on earth, checkout batteries that run on salt, and learn how liquid metal batteries can make renewable energy sources more robust.
Super-duper Fast Charging Lithium-Carbon Batteries
One of the "annoyances" relating to electric vehicles is the time it takes to charge them. Although fast-charging options exist such as the Tesla Supercharger, which can provide around 200 miles with just 15 minutes of charging, a full tank can take anywhere from six to fifteen hours to charge, which leaves some people feeling frustrated since they can't just "gas and go". What if, however, one could get a full tank/batteries within 60 to 90 seconds?
Allotrope Energy, a company based in the UK, has been developing lithium-carbon batteries that include high-rate anodes with electric double-layer capacitor-style cathodes. These batteries can be recharged in just as little as 90 seconds. These batteries provide high specific power and do not require rare earth metals. Allotrope achieves this feat by utilizing carbon, which is sourced from biomaterial, as a capacitor; this is what makes high-speed charging a possibility.
Another benefit of Allotrope’s batteries is that the same component included in “normal” lithium batteries is made out of cobalt and nickel, the sourcing of which is better for the planet and less expensive.
At present, these batteries have been used only in smaller vehicles like scooters for last-mile deliveries. However, Allotrope is working towards making the same type of batteries available in cars, imagining a future where electric cars will only need a two-minute battery recharge to travel up to 600 miles. 😊
A Battery from Mars
What if I told you there’s a battery that is as efficient as Li-ion batteries, is three times as energy dense as them and yet 90% cheaper, can store enough energy to provide a reliable backup for 100 hours, does not use rare metals that are hard to mine, and runs on CO₂?
Sounds too good to be true? Well, actually it is not. Noon Energy is developing a Carbon-Oxygen flow battery that delivers on all of these goodies.

What does this battery got to do with Mars, anyway? It is based on the same principle that is used by NASA’s MOXIE instrument to produce Oxygen from CO₂ on Mars (by the way, this is precisely what plants do back home on Earth).

In the charge cycle electricity is used to split CO₂ into Carbon Monoxide(CO) and Oxygen. The Carbon Monoxide is further reduced to solid carbon. In the discharge cycle the solid carbon combines with Oxygen to produce CO₂ and electricity. If you want to dive deeper into the nitty gritty of the battery check this article out.
This battery is best suited for energy storage at grid scale, especially in grids powered by renewable sources like wind and solar. When the wind doesn’t blow or sun doesn’t shine it can provide the necessary backup power for days at stretch.
It is not without its limitations though. It is still in the lab and it is quite bulky. It can’t fit under a car yet but perhaps it could be power a ship or a truck someday not in the not so distant future.
Salty Batteries?
Sodium-ion batteries use sodium ions to store and release energy, as opposed to lithium-ion (Li-ion) batteries that use lithium ions. What makes Na-ion batteries better than Li-ion batteries? For starters, they are cheaper to manufacture. They don’t require costly or rare materials like lithium, cobalt, or nickel. Basically, they’re made out of salt.
Na-ion batteries are safer and more environmentally friendly than Li-ion batteries. They are less likely to go into thermal runaway and combust (💥), and the materials used in Na-ion batteries are abundant and have a lower environmental impact, as compared to Li-ion batteries.
Despite these advantages, Na-ion batteries are still in the early stages of development. The current energy density of Na-ion batteries is comparable to that of contemporary lithium-iron-phosphate (LFP) batteries, which have a range of around 300 kilometers when used in Electric Vehicles. More research is needed to improve their energy density, but there is great potential for Na-ion batteries to outperform LFP batteries in the future.
To date, the majority of Na-ion battery research and development has been conducted in China. However, companies outside of China are starting to make strides in this area. Altris and Stora Enso are two companies that are conducting Na-ion battery research outside of China.
In conclusion, Na-ion batteries offer several advantages over Li-ion batteries, including cost savings, safety, and environmental friendliness. Although Na-ion batteries are still in the early stages of development and have lower energy density than Li-ion batteries, research and development in this area could potentially lead to longer-range EVs, reduce production costs, and provide an alternative to Li-ion batteries. Na-ion batteries may well be the future of EVs.
Liquid Metal Batteries
It's usually not a good thing when batteries run hot, especially when they run so hot that they can melt metals. But not so for the liquid metal batteries developed by Ambri.
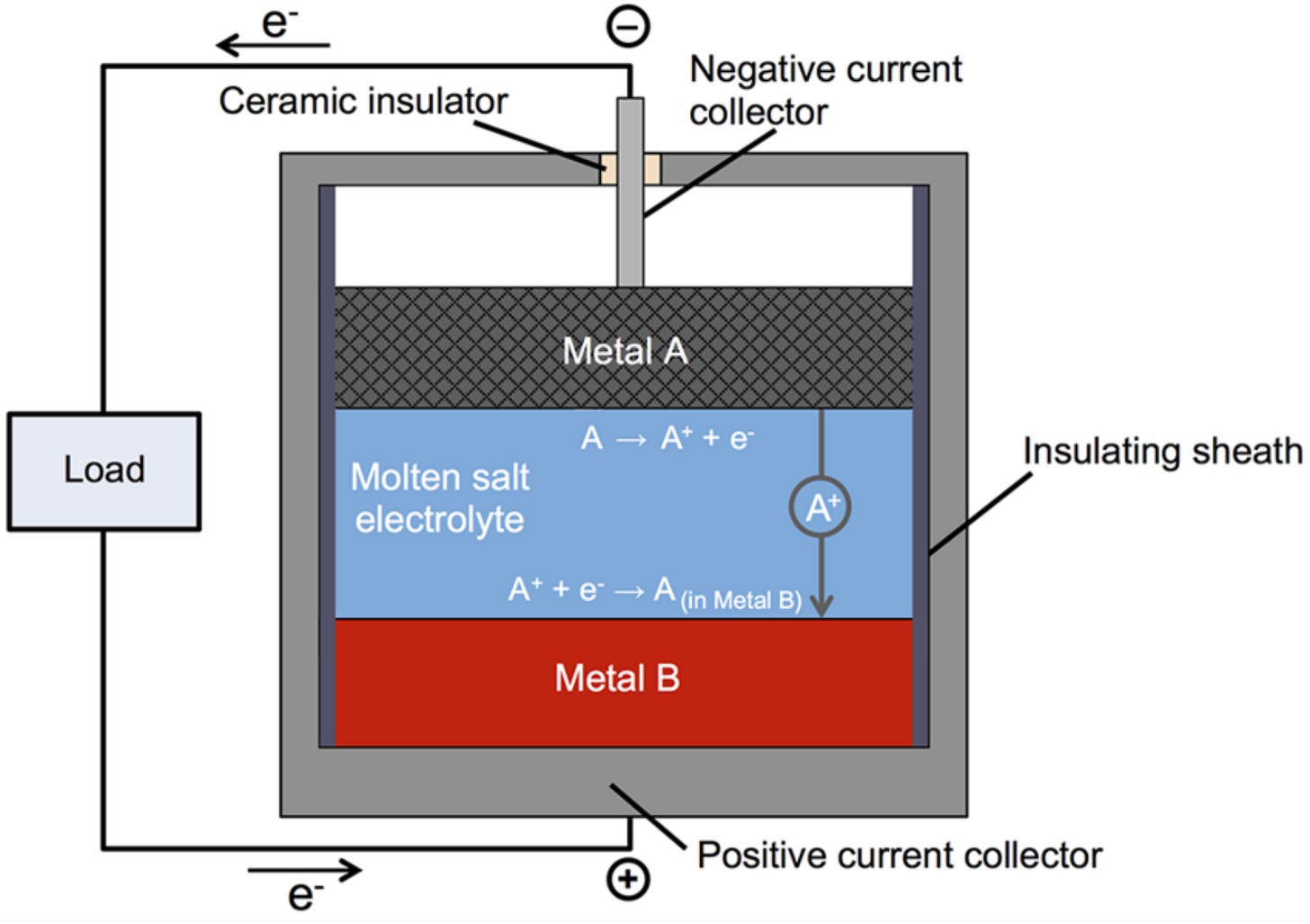
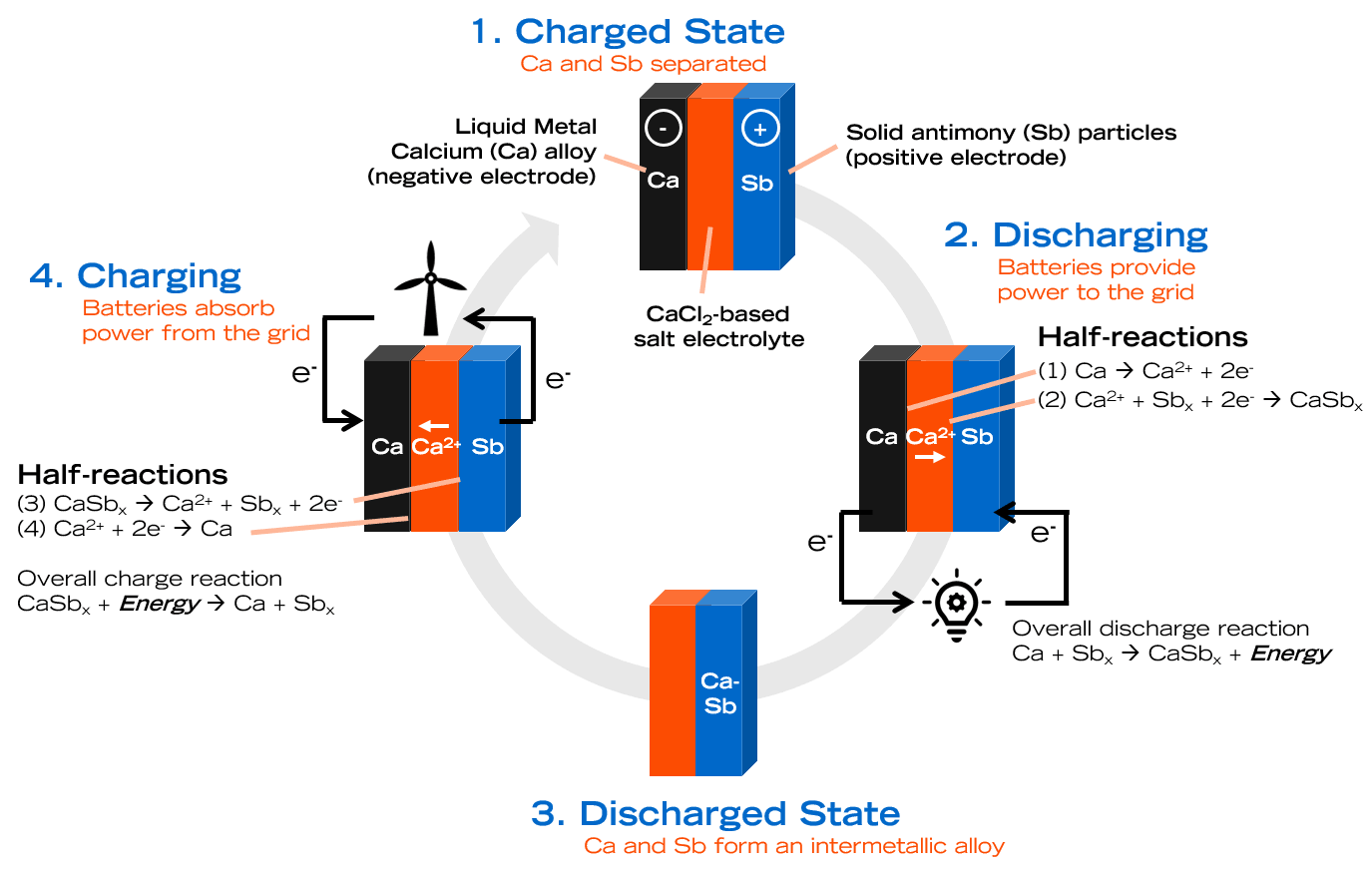
In contrast to most batteries, where the anode, cathode, and in some cases even the electrolyte are solid, a liquid metal battery has all three components in a liquid state. The anode is a low-density metal that can easily release electrons, the cathode is a high-density metal that can easily accept electrons, and the electrolyte is a molten salt that allows ions flow between the two electrodes.
Ambri's liquid metal battery uses a Calcium-alloy as the cathode, Antimony as the anode, and a calcium chloride-based salt as the electrolyte. To start the battery for the first time, its contents are heated over 500 degrees Celsius to melt the metals and electrolyte. Due to the difference in density of the three materials, they settle into distinct layers, with the Calcium-alloy at the top, Antimony at the bottom, and the electrolyte in the middle.
While discharging, the Calcium-alloy breaks down into Calcium-ions and electrons. The ions pass through the electrolyte and mix with the Antimony to form an alloy. The electrons flow through an external circuit, creating an electric current. While charging, an external current reverses this process to separate Calcium ions from the Calcium-Antimony alloy at the anode, and the separated ion passes through the electrolyte to create a Calcium-alloy at the cathode. Once the battery is operational, the heat generated by the charge and discharge cycle is sufficient to keep the components molten.
Liquid metal batteries have several advantages over Li-ion batteries. Due to their all-liquid components, they have a fast electrical response. Unlike solid electrodes, the liquid electrodes don't degrade with use, resulting in batteries that last up to 20 years. The self-separating nature of the components precludes the need for membranes, making them simpler to manufacture. The components are cheap, abundant, and easily available.
They have their fair share of limitations too. They need to be fully charged and discharged at least twice a day to maintain their operational temperature. The liquid components mean they can't be safely moved around. They also don't hold the charge for long periods. However, just like the Mars batteries, liquid metal batteries are also excellent candidates for grids powered by renewable sources, where outages drive constant discharging and charging, and immovability isn't a problem.
That’s all the charge we got for now…
If you haven’t already, check out Part 1 of the Beyond Lithium-Ion Batteries series for more of crazy battery tech. We’ll see you again in a month with exciting tech driving green innovation.
Tell a friend & Feedback
If you like this newsletter, do us a favor and share it with a friend!
If you have any feedback (or found a typo!) for us, simply hit reply. 😊